Welcome to the world of EC to PPM conversion! Dive into the depths of electrical conductivity (EC) and parts per million (ppm), unraveling their intricate relationship and exploring the diverse applications where this conversion plays a pivotal role. Our meticulously crafted EC to PPM Chart will serve as your guiding light, illuminating the path to accurate and reliable conversions.
In this comprehensive guide, we’ll delve into the factors that can influence the conversion process, ensuring you have the knowledge to navigate potential challenges. Join us as we unlock the secrets of EC to PPM conversion, empowering you with the insights to make informed decisions in various fields.
Contents
EC and PPM Units of Measurement

In various industries, measuring the concentration of substances in a solution is crucial. Electrical conductivity (EC) and parts per million (ppm) are two commonly used units of measurement for this purpose.
Electrical Conductivity (EC)
Electrical conductivity measures the ability of a solution to conduct electricity. It is expressed in units of microsiemens per centimeter (µS/cm) or millisiemens per centimeter (mS/cm). The higher the EC value, the more ions are present in the solution, and the better it conducts electricity.
Parts Per Million (PPM)
Parts per million (ppm) measures the concentration of a substance in a solution as a ratio of its mass to the total mass of the solution. It is expressed as milligrams of substance per liter of solution (mg/L). A ppm value of 1 indicates that there is one milligram of the substance dissolved in every liter of solution.
Relationship between EC and PPM
EC and PPM are related through a conversion factor that varies depending on the substance being measured. For example, the conversion factor for sodium chloride (NaCl) is approximately 0.64. This means that a solution with an EC value of 1000 µS/cm would have a ppm value of approximately 640 mg/L.
Applications of EC and PPM
EC and PPM are used in various industries to measure the concentration of substances in solutions. Some common applications include:
- Agriculture: Measuring the EC of soil and water to determine nutrient levels for crops.
- Aquaculture: Monitoring the EC of water in fish tanks to ensure optimal conditions for fish health.
- Water treatment: Measuring the EC of water to determine the presence of dissolved solids and contaminants.
- Manufacturing: Controlling the concentration of chemicals in industrial processes.
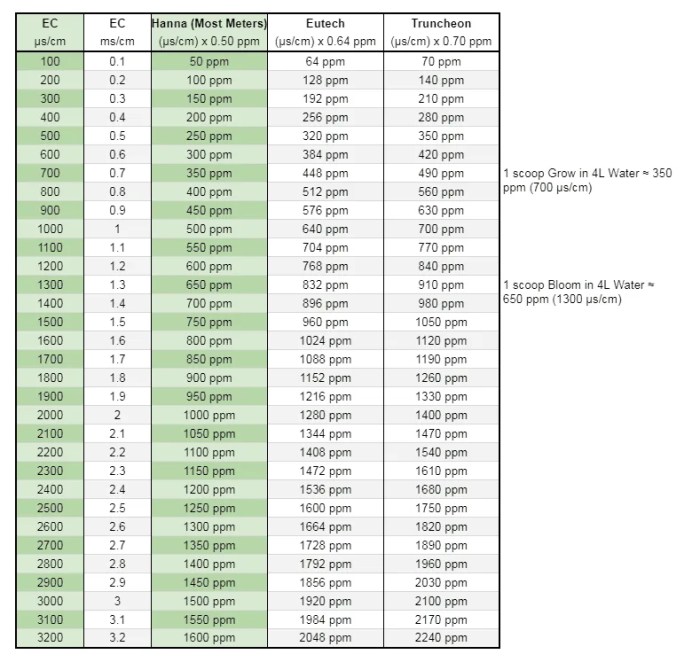
EC to PPM Conversion Chart

An EC to ppm conversion chart is a useful tool for converting electrical conductivity (EC) readings to parts per million (ppm) of dissolved solids in a solution.
EC to PPM Conversion Chart, Ec To Ppm Chart
The following table provides a conversion chart for a range of EC values and their corresponding ppm values:
| EC (mS/cm) | PPM (mg/L) |
|---|---|
| 0.1 | 50 |
| 0.2 | 100 |
| 0.3 | 150 |
| 0.4 | 200 |
| 0.5 | 250 |
| 0.6 | 300 |
| 0.7 | 350 |
| 0.8 | 400 |
| 0.9 | 450 |
| 1.0 | 500 |
To use the chart, simply find the EC value in the left column and read the corresponding ppm value in the right column.
Factors Affecting EC to PPM Conversion
The accuracy of EC to ppm conversion can be influenced by several factors, including temperature, salinity, and other variables. Understanding these factors and accounting for them when making conversions is crucial for obtaining reliable results.
Temperature
Temperature significantly affects the conductivity of a solution. As temperature increases, the mobility of ions increases, leading to higher EC readings. Conversely, lower temperatures result in lower EC readings. Therefore, it is essential to measure and adjust for temperature when converting EC to ppm.
Salinity
The presence of dissolved salts in a solution can also impact EC to ppm conversion. Different salts have varying degrees of conductivity, and their presence can affect the overall EC reading. To account for salinity, it is necessary to consider the specific types of salts present and their concentrations.
Other Variables
Other variables, such as the type of electrode used and the calibration of the EC meter, can also influence the accuracy of EC to ppm conversion. Using a high-quality electrode and regularly calibrating the EC meter ensures reliable and consistent results.
Applications of EC to PPM Conversion
EC to PPM conversion finds practical applications in various fields, including agriculture, water quality monitoring, and other industries.
Agriculture
In agriculture, EC to PPM conversion is used to determine the nutrient concentration in soil and water, which is crucial for optimal plant growth and crop yield. By measuring the EC of a soil or water sample and converting it to PPM, farmers can assess the availability of essential nutrients like nitrogen, phosphorus, and potassium. This information helps them make informed decisions regarding fertilizer application, ensuring adequate nutrient supply for plant growth.
For example, in hydroponic systems, where plants are grown in nutrient-rich water, EC monitoring is essential. By converting EC readings to PPM, growers can precisely adjust the nutrient concentration in the water to meet the specific needs of their plants.
Water Quality Monitoring
EC to PPM conversion is also used in water quality monitoring to assess the salinity and ion concentration of water sources. High EC levels can indicate the presence of dissolved salts, heavy metals, or other contaminants, which can impact water quality and pose health risks. By converting EC readings to PPM, water quality professionals can determine the suitability of water for drinking, irrigation, or industrial purposes.
For instance, in coastal areas, EC monitoring is used to track saltwater intrusion into freshwater aquifers. By measuring the EC of groundwater samples and converting it to PPM, scientists can assess the extent of saltwater contamination and its potential impact on drinking water supplies.
Limitations of EC to PPM Conversion

EC to ppm conversion provides a convenient and approximate method for estimating solute concentrations in aqueous solutions. However, it is essential to recognize its limitations and scenarios where the conversion may not yield accurate or applicable results.
Factors Affecting Conversion Accuracy
The accuracy of EC to ppm conversion is influenced by several factors:
- Solution Temperature: EC is temperature-dependent, and the conversion factor varies with temperature. This can introduce errors if the solution temperature is not accurately known or controlled.
- Solution Composition: The conversion factor is specific to the type of solutes present in the solution. Different solutes have varying ionic strengths, affecting the EC-to-ppm relationship.
- Non-Ionic Species: EC measures the electrical conductivity of ions in solution. It does not account for non-ionic species, which may contribute to the overall solute concentration but not to the EC.
Scenarios Where Conversion May Not Be Applicable
In certain scenarios, EC to ppm conversion may not be appropriate or reliable:
- Organic Solvents: EC measurement is primarily used for aqueous solutions. In organic solvents, the conversion factor may not be valid due to different dielectric constants and ionic behavior.
- Complex Solutions: In solutions with complex mixtures of solutes, the conversion factor may not accurately represent the overall solute concentration. Individual ion concentrations or alternative methods may be necessary.
- Turbid or Colored Solutions: Turbidity or color in solutions can interfere with EC measurements, affecting the accuracy of the conversion.
Alternative Methods
In cases where EC to ppm conversion is not suitable, alternative methods for measuring or estimating solute concentrations may be considered:
- Ion-Selective Electrodes (ISEs): ISEs are specific for individual ions and provide direct measurements of their concentrations.
- Gravimetric Analysis: This method involves evaporating a known volume of solution and measuring the mass of the remaining solute.
- Titration: Titration techniques use chemical reactions to determine the concentration of specific solutes.
Understanding the limitations of EC to ppm conversion and employing appropriate alternative methods when necessary ensures accurate and reliable measurements of solute concentrations in various solutions.
Epilogue: Ec To Ppm Chart

As we conclude our exploration of EC to PPM conversion, we hope you’ve gained a deeper understanding of this essential process. Remember, the EC to PPM Chart is your trusty companion, ready to assist you in your endeavors. Whether you’re navigating the intricacies of agriculture, unraveling the mysteries of water quality, or venturing into other industries, this conversion tool will be your steadfast guide.
Expert Answers
What is the significance of EC to PPM conversion?
EC to PPM conversion plays a crucial role in various industries, including agriculture, water quality monitoring, and environmental science. It allows us to quantify the concentration of dissolved solids in a solution, providing valuable insights into nutrient availability, water purity, and overall environmental health.
Can EC to PPM conversion be affected by external factors?
Yes, EC to PPM conversion can be influenced by factors such as temperature, salinity, and the presence of specific ions in the solution. It’s essential to consider these factors when performing conversions to ensure accurate results.
What are the limitations of EC to PPM conversion?
While EC to PPM conversion is a valuable tool, it’s important to recognize its limitations. In certain cases, the conversion may not be accurate due to the presence of non-ionic compounds or complex interactions within the solution. Alternative methods may be necessary in such scenarios.