11+ Which Intervals Are Affected By The Addition Of A Catalyst. Automobile catalytic converters use a platinum catalyst to reduce air pollution by changing emissions such as carbon monoxide, co(g), into carbon dioxide, co 2 (g). There will be two cases, i.e.
How are the following aspects of a reaction affected by the addition of a catalyst? (1) 1 and 2 (2) 1 and 3 (3) 2 and 4 (4) 3and 4. How are the following aspects of a reaction affected by the addition of a catalyst.
Contents
- 1 A Catalyst Lowers The Activation Energy, For Example, By.
- 2 One Is The Addition Of A Catalyst Is Positive, And The Other Is The Addition Of A Catalyst Is Negative.
- 3 Adding A Catalyst Will Increase The Rate Of Reaction Significantly.
- 4 They Are Not Used Up In The Reaction.
- 5 Although Catalysts Have No Effect On The Position Of Equilibrium,I.e.the Yield Of The Reaction, They Do Allow Equilibrium To Be Reached More Quickly.
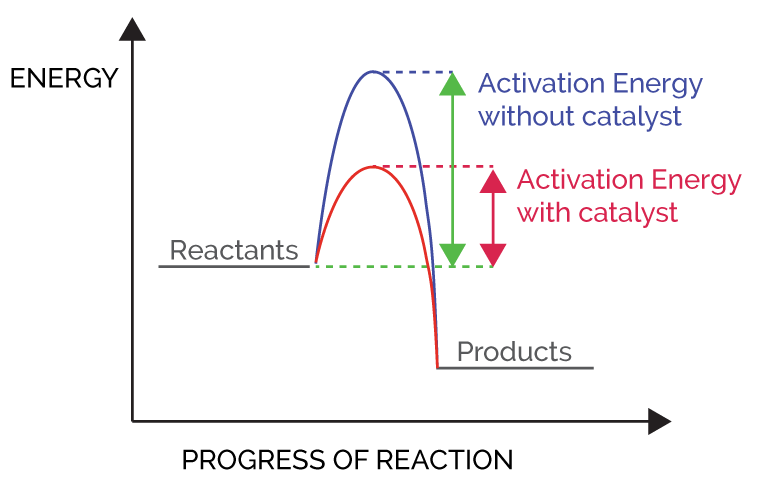
A Catalyst Lowers The Activation Energy, For Example, By.
Which numbered interval represents the heat of reaction? From the basic chemistry it is known catalysts are substance that accelerates the rate of. How are the following aspects of a reaction affected by the addition of a catalyst.
One Is The Addition Of A Catalyst Is Positive, And The Other Is The Addition Of A Catalyst Is Negative.
Exothermic mg (s) + 2agno3. Thus, activation energy is being affected by the catalyst. How are the following aspects of a reaction affected by the addition of a catalyst?
Adding A Catalyst Will Increase The Rate Of Reaction Significantly.
Increased decreased not affected rate of the forward reaction activation energy of the reverse reaction. A)a catalyst b)an indicator c)electrical energy d)thermal energy 5.the activation energy of a chemical reaction can be. Rate of reaction can be increased by using a catalyst.
They Are Not Used Up In The Reaction.
There will be two cases, i.e. Reactions can be sped up by the addition of a catalyst, including reversible reactions involving a final equilibrium state. Anything that touches the top of the hill:
Although Catalysts Have No Effect On The Position Of Equilibrium,I.e.the Yield Of The Reaction, They Do Allow Equilibrium To Be Reached More Quickly.
45 which balanced equation represents a. Verified by toppr it increases the rate of reaction. In the presence of a catalyst, both the forward and reverse reaction rates will speed up equally, thereby allowing the system to.